AbstractBackgroundChloride-rich fluid administration is frequently employed in the management of aneurysmal subarachnoid hemorrhage (aSAH). However, the incidence and consequences of hyperchloremia in aSAH remain poorly defined. This study aimed to describe the incidence of hyperchloremia in aSAH, the contribution of fluid sources to chloride exposure, and the potential associations of hyperchloremia with patient outcomes.
MethodsThis was a single-center retrospective cohort study of patients admitted to a neurointensive care unit with aSAH. The primary outcome was incidence of hyperchloremia (chloride >109 mEq/L). Secondary outcomes included incidence of severe hyperchloremia (chloride >115 mEq/L), incidence of acute kidney injury (AKI), need for renal replacement therapy (RRT), intensive care unit (ICU) length of stay (LOS), hospital LOS, and in-hospital mortality.
ResultsOf the 234 patients included in the analysis, hyperchloremia occurred in 75% (n=175), and 58% (n=101) developed severe hyperchloremia. Median time to onset was 3 days (interquartile range, 1–5) after admission. Hyperchloremia was associated with prolonged ICU LOS (12 vs. 8 days, P<0.001), duration of mechanical ventilation (16 vs. 10 days, P<0.001), hospital LOS (15 vs. 9 days, P<0.001), and in-hospital mortality (14.3% vs. 0%, P=0.002) compared to no hyperchloremia. No significant difference was observed in the incidence of AKI or the need for RRT. Maintenance intravenous fluids accounted for the highest proportion of the cumulative chloride burden.
INTRODUCTIONAneurysmal subarachnoid hemorrhage (aSAH) is a neurosurgical emergency with an estimated mortality rate of 20% [1]. Among survivors, nearly 45% suffer from long-term disabling side effects [2]. The neurologic consequences associated with aSAH including delayed cerebral ischemia (DCI), cerebral vasospasm, and cerebral edema portend a poor prognosis and carry a high degree of morbidity and mortality. Aside from the direct neurologic sequelae, patients with aSAH are at a higher risk of developing secondary non-neurologic complications including cardiac dysfunction, infection, hepatic and renal failure, which negatively impact patient outcomes [3]. Despite the contemporary shift away from hypervolemia, as historically targeted in so-called Triple-H therapy, fluid administration to maintain euvolemia remains a cornerstone in the management of aSAH to mitigate the effects of DCI, prevent secondary brain injury, and optimize organ perfusion [4-6].
Furthermore, the selection of optimal fluid type in aSAH treatment remains ill-defined and lacks standardization. It is well understood that hypertonic or isotonic chloride-rich fluids such as hypertonic saline or normal saline are most commonly employed due to the perceived risk of cerebral swelling with hypotonic fluids [4]. However, hyperchloremia secondary to chloride-rich fluid administration has been linked with adverse consequences, including acute kidney injury (AKI), acid-base disturbances, and a prolonged need for mechanical ventilation in critically ill patients [7]. Moreover, in animal models, hyperchloremia has been shown to result in immunosuppression, increased presence of inflammatory markers, and a subsequent risk of infection [8]. Although the risks of fluid therapy have been well characterized in medically critically ill patients, the risks in neurocritically ill patients with aSAH remain to be elucidated.
While patients with aSAH receive a majority of the fluid intake from intravenous (IV) bolus or maintenance IV infusions, there are various hidden sources of chloride-rich fluids in the form of enteral fluids and medication diluents such as intermittent IV infusions (so-called piggybacks; for example, antibiotics, antiseizure medications) and/or titrated continuous infusion (for example, sedatives and vasoactive medications). Hidden obligatory fluid intake from medications/diluents is often an unrecognized yet significant contribution to the overall fluid balance [9]. Observational studies have shown that patients receive high volumes of crystalloid administration, with an estimated five liters or more in the acute phase of aSAH and only a modest reduction to a minimum of three liters by day 13 of hospitalization [10]. Characterizing the source and contribution of this significant volume of cumulative fluid intake in patients with aSAH will not only increase awareness of cumulative fluid burden but also assist in identifying key areas for implementation of fluid stewardship interventions.
This retrospective, single-center, cohort study aimed to describe the incidence of hyperchloremia in patients with aSAH and the contribution of fluids, medication diluents, and other fluid sources to chloride administration. We hypothesized that hyperchloremia, defined as a serum chloride level >109 mEq/L, occurs at a high incidence in patients with aSAH due to excessive chloride-rich fluid intake, and is associated with worse patient outcomes, such as AKI, length of stay (LOS), and mortality.
METHODSSetting and participantsThis was a retrospective, single-center, cohort study of patients admitted to the neurointensive care unit with a diagnosis of aSAH between January 1, 2015, and August 31, 2019. Patients between 18 and 85 years were enrolled if they had an index International Classification of Diseases, 9/10 diagnosis code for aSAH, received oral nimodipine, and were admitted to the neurointensive care unit for longer than 48 hours. The receipt of oral nimodipine was chosen to ensure a homogeneous patient population with an index admission for acute spontaneous aSAH. Patients were excluded if they were diagnosed with traumatic SAH, pregnant, or incarcerated; had AKI or chronic kidney disease (CKD) requiring renal replacement therapy (RRT) on admission; were moribund; or had early withdrawal of care.
Study outcomes and definitionsThe primary outcome of the study was the incidence of hyperchloremia, defined as a serum chloride level >109 mEq/L. [11] Secondary outcomes included the incidence of severe hyperchloremia >115 mEq/L, incidence of AKI, need for RRT, characterization of the source of fluid intake (mL/day) and chloride intake (mEq/day), length of intensive care unit (ICU) stay (days), length of hospital stay (days), and in-hospital mortality.
Data collectionData were collected from the University of Kentucky electronic health record and included baseline demographics, pre-existing comorbidities, pertinent laboratory values, the presence of cerebral vasospasm, assessment of renal function (serum creatinine, urine output, need for RRT), daily IV medication/fluids, enteral sodium tablets/solution received during ICU LOS up to day 14, nephrotoxin exposure, daily total daily fluid intake and output, length of hospital/ICU stay, and mortality. AKI was defined according to the Kidney Disease: Improving Global Outcomes (KDIGO) criteria as stage 1 and above [12]. Data regarding the Hunt Hess score and modified Fisher grade were individually collected by the investigator team. Patient data were evaluated for the duration of admission, and no follow-up or data collection was performed after discharge.
To quantify the contribution of hyperchloremia from medications, daily data on IV medications administered including diluent, volume, dose, and frequency were obtained. Data collection on fluid intake was limited to 14 days due to the decline in sample size after 14 days and because the first 14 days portend the highest risk for vasospasm and DCI in patients with aSAH, thus constituting the period with the most aggressive fluid supplementation. Potential nephrotoxin exposure was defined as receipt of any of the following medications: vancomycin (IV), amphotericin (IV), aminoglycosides (IV), acyclovir (IV), angiotensin-converting enzyme inhibitors (IV/ oral [PO]), angiotensin receptor blockers (PO), nonsteroidal anti-inflammatory medications (IV/PO), piperacillin-tazobactam (IV), loop diuretics (IV/PO), contrast (IV), and trimethoprim-sulfamethoxazole (IV/PO).
Statistical analysisThe a priori statistical analysis plan included dividing the cohort into those who met the primary outcome of hyperchloremia and comparing the baseline characteristics and outcomes to those who did not develop hyperchloremia. No sample-size calculations were performed. Data are presented as means with standard deviations (SDs) or medians with interquartile ranges (IQRs) depending on their underlying distributions. Continuous variables were analyzed using either Student t-test or the Mann-Whitney U-test. Categorical variables were analyzed using Pearson’s chi-square or Fisher’s exact test as appropriate and were reported as counts with percentages. The missing values were not imputed.
Multivariable regressionMultivariable logistic regression was used to model the primary outcome of hyperchloremia and secondary outcome of AKI. For the primary outcome, variables found to be significant in the bivariate analysis were selected for the hyperchloremia model and included basic demographic variables (age, sex), Sequential Organ Failure Assessment (SOFA) score, Hunt-Hess score, modified Fisher grade, cumulative fluid intake, baseline serum chloride concentration, mean daily chloride dose, and chloride dose within the first 48 hours of admission). Given the wide degree of variability in the volume of fluid administration and intake during the study period, cumulative fluid intake was assessed as a continuous variable and divided into quartiles (<25%, 25%–50%, 51%–75%, and >75%) to improve interpretation. The primary independent variable during the study period was hyperchloremia. A backward elimination selection criterion was used at a level of 0.05. Bivariate analysis was performed using statistical software, IBM SPSS version 27 (IBM Corp., Armonk, NY, US), and multivariable modeling was performed using SAS version 9.4 (SAS Institute, Cary, NC, USA). A multivariable logistic regression model was also used to identify predictive factors for AKI using the same procedures. The variables selected for this model were a priori and included sex, age, Hunt-Hess score, Fisher grade, development of hyperchloremia, CKD, sepsis, and nephrotoxin exposure at 48 hours.
RESULTSDemographicsA total of 234 patients with aSAH admitted to the ICU over a four-year period met the inclusion criteria for the primary analysis. Baseline demographics were similar between groups. Most of the cohort was female sex (59%), with a mean age of 55 years (SD, 13). Hypertension (79.5%) and diabetes (22.2%) were the most common preexisting comorbidities. A total of 4% of the population had CKD at baseline, and none of the patients required RRT. The median Hunt-Hess score and modified Fisher scale on admission were 2 (IQR, 1–4) and 3 (IQR, 2–4), respectively. The mean SOFA score was 5.6 (SD, 3.8). The median length of ICU stay for the entire cohort was 10 days, corresponding to a median total hospital stay of 13 days. The overall mortality during hospital admission in this population was 11%. A full description of the baseline characteristics of both cohorts is provided in Table 1.
Primary outcomeThe primary outcome of the incidence of hyperchloremia (chloride >109 mEq/L) was 75% (n=175) of the study population, with 58% (n=101) developing severe hyperchloremia (chloride >115 mEq/L). The median chloride in patients who developed hyperchloremia was significantly higher at 115 mEq/L (IQR, 112–120 mEq/L) compared to 108 mEq/L (IQR, 106–109 mEq/L) in those who did not develop hyperchloremia (P<0.001) (Table 2). The median time to the onset of hyperchloremia was 3 days (IQR, 1–5 days) after admission. Females were more likely to have hyperchloremia than males (64% vs. 36%; odds ratio [OR], 2.4; 95% confidence interval [CI], 1.3–4.4). No differences in the incidence of hyperchloremia were associated with race or ethnicity (P=0.59). The Hunt-Hess score and modified Fisher grade were both significantly associated with an increased incidence of hyperchloremia (Table 1), although there was no significant difference. Chloride concentrations increased to the maximum mean concentration on day 4 in patients with hyperchloremia (day 2 in those without hyperchloremia) and were consistently sustained throughout the first 14 days of admission (Fig. 1).
Secondary outcomesThe development of hyperchloremia was associated with prolonged length of ICU stay (12 vs. 8 days, P<0.001), duration of mechanical ventilation (16 vs. 10 days, P<0.001), longer length of hospital stay (15 vs. 9 days, P<0.001), and a higher rate of hospital mortality (14.3% vs. 0%, P=0.002) than in patients who did not develop hyperchloremia. The rates of AKI were similar in the two groups (4% vs. 5.1%, P=0.716; OR, 2.1; 95% CI, 0.6–7.5), with no further difference in KDIGO stage or the need for RRT (Table 2). The mean serum chloride level by hospital day also did not differ between patients with and without AKI (Fig. 1).
Multivariate regressionIn the multivariable logistic regression analysis for hyperchloremia when accounting for SAH severity, age, and chloride dose within the first 48 hours, variables such as sex, higher admission SOFA score, cumulative fluid intake, serum chloride concentration on admission, and daily mean chloride dose were independently predictive of hyperchloremia (Table 3). For every 1 liter increase in chloride-rich fluid intake, the risk of hyperchloremia increased by 4% (OR, 1.04; 95% CI, 1.01–1.06). Male sex and daily chloride dose were inversely associated with the development of hyperchloremia.
In the multivariate logistic regression model for AKI, accounting for sex, age, Hunt-Hess score, Fisher grade, development of hyperchloremia, CKD, sepsis, and nephrotoxin exposure at 48 hours, the presence of CKD, sepsis, and nephrotoxin exposure were all independent predictors of AKI (Supplementary Table 1).
Fluid intakeWhile there was a statistically significantly higher median cumulative fluid intake over the first 14 days in patients who developed hyperchloremia (47.6 L vs. 26.6 L, P<0.0001), there was no difference in the mean daily or median cumulative chloride exposure within the first 72 hours of hospital stay. Serum chloride mirrored daily fluid intake and overall daily fluid balance throughout the first 14 days of admission (Fig. 2). Overall, patients received a mean daily fluid intake of 3.7 liters between days 1 and 7, with only a small reduction to 3.6 liters on days 8 to 14. Maintenance IV fluids contributed to the highest volume, chloride content, and sodium content per day. Patients with hyperchloremia had a median daily chloride dose of 334 mEq (IQR, 150–683 mEq) compared to 405 mEq (IQR, 233–789 mEq) in patients with no hyperchloremia (P=0.228). In the overall cohort, the mean sodium and chloride dose per day was 636.5 mEq (SD, 112) and 646 mEq (SD, 113), respectively.
Over the course of the study period, the majority of fluid intake was from enteral nutrition (47%), maintenance IV fluids (19%), and titrated IV infusions (18%) (Fig. 3). The majority of fluid intake came from maintenance IV fluids early in the study period (day 1, 57.5%) but reduced in proportion over the length of the study period (Supplementary Fig. 1A). The amount of chloride administered was primarily from maintenance IV fluids (overall 48.1%) (Supplementary Fig. 1B). Enteral sodium supplementation and nutrition (31%) and IV boluses (12%, typically 3% sodium chloride) also substantially contributed to the daily chloride dose. Over the first 14 days, the percentage of chloride delivered each day shifted from maintenance IV fluids (67.1% of all chloride provided) to an almost equal split of maintenance IV fluids (43.2%) and enteral sodium supplementation (42.1%, usually sodium chloride tablets) (Supplementary Fig. 1B).
DISCUSSIONIn the current observational study of 234 patients with aSAH, a high incidence of hyperchloremia (75%) was observed. Elevated serum chloride concentrations were associated with the volume of maintenance IV infusion but not with the dose of chloride administered. While the overall outcomes of the population were similar to those expected in the aSAH population, the development of hyperchloremia was associated with worse outcomes related to prolonged duration of ICU/hospital LOS, prolonged duration of mechanical ventilation, numerically higher rates of AKI, and an increased risk of in-hospital mortality. The need for mechanical ventilation, higher SOFA score, and presence of sepsis likely contributed to the indication for increased fluid intake, resulting in hyperchloremia, and was associated with worsened outcomes in this study.
Hyperchloremia has been demonstrated to be strongly associated with AKI in critically ill patients admitted to the medical ICU [7]. In the current study, AKI occurred in 10.3% of patients overall and was not significantly associated with the occurrence of hyperchloremia. The presence of sepsis, CKD, and nephrotoxin exposure at 48 hours was found to be an independent predictive factor for AKI, similar to previous studies evaluating the risk factors for AKI in neurologically injured patients [13]. In a retrospective analysis by Sadan et al. [1], every 10 mEq/L increase in serum chloride level was associated with a 7.39 increase in the odds of AKI among patients with aSAH (OR, 7.39; 95% CI, 3.44–18.23). Potential factors that may have contributed to the lower incidence of AKI in this population were the small population cohort and the detection of AKI based on serum creatinine levels alone.
Previous studies have identified that medication diluents contribute to the highest volume and chloride intake among critically ill patients admitted to the medical ICU [9]. Normal saline (0.9% sodium chloride) has been used as the primary diluent for most medications and was identified to independently increase the risk of hyperchloremia and subsequent AKI compared with substitution with a dextrose-containing diluent [9]. The implications of the findings by Magee et al. [9] and those of the current study suggest a potential need for intervention in substituting medication diluents as a mitigation strategy to prevent hyperchloremia. However, dextrose-containing fluids are often avoided in patients with acute neurologic injuries because of the risk of exacerbating cerebral edema or promoting hyponatremia. Potential therapeutic alternatives to normal saline to mitigate chloride exposure and avoid the use of hypotonic fluids may include the use of sodium acetate or mixed sodium acetate/chloride solution (that is, “buffered saline”) [14].
The recently published ACETatE trial (A Low-Chloride Hypertonic Solution For Brain Edema) was a randomized pilot trial assessing the efficacy of 23.4% sodium chloride vs a mixture of 16.4% sodium chloride/sodium acetate for the treatment of cerebral edema. The trial demonstrated a lower chloride load and incidence of AKI in patients who had received a mixed saline/acetate solution compared to sodium chloride solution alone (11.8% vs. 53.3%, P=0.01) [11]. This study supports the hypothesis that excess chloride load may be a potentiator of AKI in this population and that interventions targeted at mitigating chloride load may preserve renal function among patients with aSAH. The current study found that the highest proportion of the chloride load came from the maintenance of IV fluids. Ongoing studies are needed to assess the safety and feasibility of substituting other sodium salts with sodium chloride as a maintenance IV fluid or reducing the use of hypertonic saline and targeting high serum sodium concentrations [15].
The limitations of the current study include its retrospective observational design. While the data extracted from the electronic health record were validated by selected manual confirmation, documentation of fluids in and out daily is subject to some natural variability in the course of patient care (in particular, oral consumption that may go unmeasured). However, great attention has been paid to the fluid balance of these patients after aSAH, so there is confidence that the documentation is as precise and accurate as is reasonable to expect for a non-prospective study scenario. Owing to inconsistencies in documentation, the incidence of vasospasm could not be accurately assessed in all patients based on retrospective data collection. The presence of vasospasm would be predicted to result in a higher volume of fluid administration to optimize cerebral perfusion, and thus, a higher cumulative chloride load. Patients with vasospasm may also receive more IV contrast agents or have a higher systemic inflammatory state, both of which could increase the risk of AKI. The use of serum creatinine as a marker of kidney function has known limitations, and its use for the diagnosis of AKI may have limited accuracy. Thus, the incidence of AKI may be underreported [16]. Further studies should evaluate AKI using more accurate biomarkers of kidney injury in this population.
In this retrospective observational cohort study of 234 patients with aSAH, the incidence of hyperchloremia was 75%. Hyperchloremia was not associated with a statistically significant increased risk of AKI; however, patients who developed hyperchloremia experienced worse outcomes such as prolonged ICU LOS, hospital LOS, duration of mechanical ventilation, and mortality. Maintenance IV fluids, followed by bolus and continuous IV fluids, contributed to the highest chloride burden. Awareness of and adjustments to the chloride load in maintenance IV fluids and medication diluents may assist in reducing the chloride burden in this patient population as well as the risk of hyperchloremia. Further prospective studies are needed to assess the impact of reducing the chloride burden and the incidence of hyperchloremia in patients with aSAH on the improvement of clinically important patient outcomes.
ARTICLE INFORMATIONSupplementary materialsSupplementary materials can be found via https://doi.org/10.18700/jnc.220068.
Supplementary Fig. 1.Individual contributors to fluid intake. Average total volume of fluid administration (A) and total chloride dose (B) per day stratified by type/source of fluid administered over the 14-day study period. Day 0 was defined as date of hospital admission. IV, intravenous; IVPB, intravenous piggyback; MIVF, maintenance intravenous fluid. Fig. 3.Total fluid volume (A) and chloride dose (B) received, by category. IV, intravenous; IVPB, intravenous piggyback; MIVF, maintenance intravenous fluid. 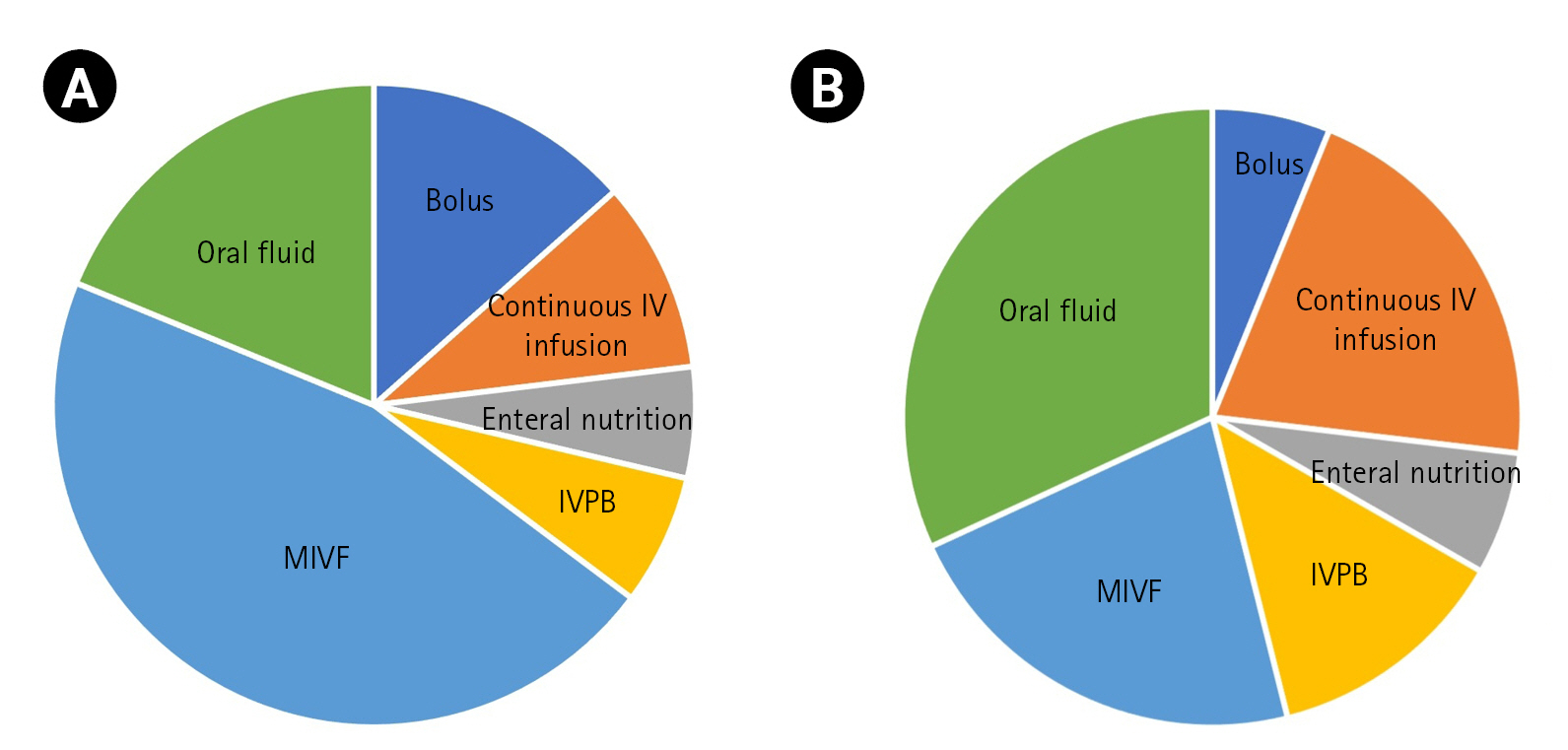
Table 1.Baseline patient characteristics (n=234)
Table 2.Secondary outcomes
Table 3.Multivariable logistic regression model for hyperchloremia
REFERENCES1. Sadan O, Singbartl K, Kandiah PA, Martin KS, Samuels OB. Hyperchloremia is associated with acute kidney injury in patients with subarachnoid hemorrhage. Crit Care Med 2017;45:1382-8.
2. Patel VN, Samuels O. The critical care management of aneurysmal subarachnoid hemorrhage. In: Murai Y, editor. Aneurysm. Rijeka: IntechOpen; 2012.
3. Wartenberg KE, Schmidt JM, Claassen J, Temes RE, Frontera JA, Ostapkovich N, et al. Impact of medical complications on outcome after subarachnoid hemorrhage. Crit Care Med 2006;34:617-23.
4. van der Jagt M. Fluid management of the neurological patient: a concise review. Crit Care 2016;20:126.
5. Connolly ES Jr, Rabinstein AA, Carhuapoma JR, Derdeyn CP, Dion J, Higashida RT, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2012;43:1711-37.
7. Neyra JA, Canepa-Escaro F, Li X, Manllo J, Adams-Huet B, Yee J, et al. Association of hyperchloremia with hospital mortality in critically ill septic patients. Crit Care Med 2015;43:1938-44.
8. Payen D, Haloui H. Acid-base status is an important factor for inflammation, but don’t forget CO2! Crit Care 2014;18:664.
9. Magee CA, Bastin MLT, Laine ME, Bissell BD, Howington GT, Moran PR, et al. Insidious harm of medication diluents as a contributor to cumulative volume and hyperchloremia: a prospective, open-label, sequential period pilot study. Crit Care Med 2018;46:1217-23.
10. Rass V, Gaasch M, Kofler M, Schiefecker AJ, Ianosi BA, Steinkohl F, et al. Fluid intake but not fluid balance is associated with poor outcome in nontraumatic subarachnoid hemorrhage patients. Crit Care Med 2019;47:e555-62.
11. Sadan O, Singbartl K, Kraft J, Plancher JM, Greven AC, Kandiah P, et al. Low-chloride- versus high-chloride-containing hypertonic solution for the treatment of subarachnoid hemorrhage-related complications: the ACETatE (a low ChloriE hyperTonic solution for brain Edema) randomized trial. J Intensive Care 2020;8:32.
12. Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 2012;120:c179-84.
13. Büttner S, Stadler A, Mayer C, Patyna S, Betz C, Senft C, et al. Incidence, risk factors, and outcome of acute kidney injury in neurocritical care. J Intensive Care Med 2020;35:338-46.
14. Suarez JI, Qureshi AI, Parekh PD, Razumovsky A, Tamargo RJ, Bhardwaj A, et al. Administration of hypertonic (3%) sodium chloride/acetate in hyponatremic patients with symptomatic vasospasm following subarachnoid hemorrhage. J Neurosurg Anesthesiol 1999;11:178-84.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||