Failed recanalization mediates the association of women with poor outcomes after thrombectomy: a single-center experience
Article information
Abstract
Background
Whether thrombectomy benefits differ according to sex remains debatable. We aimed to investigate whether there was a difference in stroke outcomes between men and women treated with thrombectomy.
Methods
We studied 173 patients with anterior circulation strokes. Failed recanalization was defined as thrombolysis in cerebral infarction grade 0-2a. Scores >2 on the modified Rankin Scale at 3 months were regarded as poor outcomes. To prove that failed recanalization mediated the association between sex differences and functional outcome, the four steps of the reasoning process adapted from Baron and Kenny’s causal-steps approach were tested. The adjusted odds ratios (aORs) and 95% confidence intervals (CIs) were calculated.
Results
This study included 76 women and 97 men. Women were older and presented with atrial fibrillation more frequently than men. Female sex was independently associated with failed recanalization (aOR, 2.729; 95% CI, 1.334–5.582), which was an independent predictor of poor outcomes (aOR, 4.630; 95% CI, 1.882–11.389). Women were associated with poor outcomes in the analysis adjusted for confounders, except for failed recanalization (aOR, 2.285; 95% CI, 1.064–4.906). However, the association became insignificant in the additional analysis adjusted for failed recanalization (aOR, 1.670; 95% CI, 0.738–3.784). The indirect effect between female sex and poor outcomes via failed recanalization was statistically significant (aOR, 1.038; 95% CI, 1.010–1.127).
Conclusion
Our study showed that failed recanalization mediated the association between women and poor outcomes after thrombectomy. Nonetheless, this might be explained by chance given our limited study population.
INTRODUCTION
Mechanical thrombectomy has recently been established as an essential treatment modality for emergent large vessel occlusion within 8–24 hours of symptom onset, as multiple randomized clinical trials (RCTs) have demonstrated its dramatic efficacy in the improvement of patient outcomes [1-5]. Using the data from the RCTs, some studies reported that the benefit of thrombectomy was equal in both sexes [6,7], while other studies showed differences in the outcomes between men and women treated with thrombectomy [8,9]. On the other hand, several studies using nonclinical trial data investigated whether the benefit of thrombectomy differed by sex. However, the results are conflicting among such studies [10-12], and thus this topic is still debatable. In addition, real-world nonclinical trial data to date are insufficient in the literature. Thus, in this study, using single-center nonclinical trial data, we aimed to investigate whether there is a significant difference in clinical outcomes between men and women treated with thrombectomy.
METHODS
Patient recruitment
We retrospectively reviewed the medical records of 216 patients who were prospectively enrolled in the patient registry for endovascular thrombectomy at Soonchunhyang University Bucheon Hospital from January 2012 to November 2020. Among them, we excluded 20 patients with posterior circulation stroke, 16 patients without collateral score data (no computed tomography [CT] angiography performed), four patients without outcome data, and three patients who proved to have cancer-related strokes. Finally, 173 patients with anterior circulation stroke were included in this study.
Patient treatment process
For all the included patients, non-contrast-enhanced CT and CT angiography were performed before treatment. Using a 128-detector high-definition CT scanner (Discovery CT750 HD; GE Healthcare, Milwaukee, WI, USA), CT angiography images were obtained from the aortic arch to the vertex in series (section thickness, 0.625 mm; tube voltage, 100 kV; tube current, 200 mA) after a single bolus injection of 100 mL nonionic contrast agent into the antecubital vein. Candidates for intravenous thrombolysis (IVT) and thrombectomy were selected in accordance with previously published guidelines [13,14]. Notably, from November 2019, patients with stroke within 6 to 24 hours of the last known normal were also considered to be treated by thrombectomy if a mismatch between the infarct core and penumbra—the presence of a significant salvageable tissue—was confirmed on CT perfusion images [4,5].
Catheter angiography for thrombectomy was performed via the femoral artery under local anesthesia. If necessary, conscious sedation was used at the discretion of the treating interventionists. An 8-Fr flow-gate balloon guide catheter was placed in the proximal internal carotid artery to approach the target artery. Thrombectomy was performed using a stent retriever (Solitaire, Medtronic, Dublin, Ireland or Trevo, Stryker, Kalamazoo, MI, USA) or an aspiration device (Penumbra system; Penumbra Inc., Alameda, CA, USA). Non-contrast-enhanced CT and magnetic resonance diffusion-weighted imaging (DWI) were performed immediately and 24 hours after the procedure, respectively. Additional CT or DWI was performed on the basis of the decision of the treating physician.
Clinical data and imaging analysis
The following clinical information was obtained: age, sex, hypertension, diabetes mellitus, hyperlipidemia, history of prior stroke, ischemic heart disease, atrial fibrillation, current smoking habits, premorbid independence (pre-stroke modified Rankin Scale [mRS] ≤2), history of antithrombotic and statin medication use, initial blood pressure, blood glucose level, National Institutes of Health Stroke Scale (NIHSS) score, time interval from stroke onset to arrival at the emergency department and from arrival to groin puncture, thrombectomy procedure time, location of symptomatic occlusion, clot burden score [15], use of IVT, and stroke classification [16].
The Alberta Stroke Program Early CT score (ASPECTS) was rated based on pretreatment CT angiography source images (CTA-SI). All images were adjusted to have maximum contrast between the normal and abnormal sides. Diminished contrast enhancement in each region of ASPECTS compared with the healthy hemisphere was considered abnormal [17]. In addition, collateral status was dichotomized as poor (≤50% filling of the occluded territory) or good (>50% filling) status [18]. The final recanalization state was assessed using catheter angiography based on the thrombolysis in cerebral infarction (TICI) perfusion scale after the thrombectomy procedure was finished [19]. Failed recanalization was defined as a TICI grade of 0-2a. Hemorrhagic transformation and infarct size were determined on the basis of follow-up gradient-echo images and DWIs. Symptomatic intracranial hemorrhage [20], hemorrhagic infarct, and parenchymal hemorrhage [21] were defined according to the previously published criteria. All images were analyzed independently by two investigators (SJL and TKL) blinded to the clinical data. The final decision was made by consensus. The 3-month mRS score was documented in the follow-up clinic note by a neurologist or neurosurgeon. Scores of ≤2 (>2) on the mRS at 3 months were regarded as good (poor) functional outcomes.
Statistical analysis
Statistical analyses were performed using IBM SPSS version 21.0 (IBM Corp., Armonk, NY, USA) and R software, version 4.0.5 (The R Foundation for Statistical Computing, Vienna, Austria). Univariate group comparisons were performed using the independent two-sample t-test (or Mann-Whitney U-test for non-normally distributed continuous variables) and the chi-square test (or Fisher’s exact test). The assumptions of normality and equivalent variance were assessed using the Shapiro-Wilk test and Levene's test, respectively. Age and variables with P<0.1 in the univariate comparison were included in the multivariable logistic regression analysis for failed recanalization and poor functional outcomes. Procedural time was excluded from the multivariable logistic analysis for functional outcomes because it was associated with failed recanalization (P=0.006 by independent t-test). To prove that failed recanalization mediated the association between sex differences and functional outcome, we tested the four steps of the reasoning process adapted from Baron and Kenny’s causal-steps approach [22,23]. Specifically, using adjusted logistic regression, we attempted to prove the following steps: (1) the significant association of women (predictor) with failed recanalization (dependent variable), (2) the significant association of failed recanalization (predictor) with poor outcomes (dependent variable), (3) the significant association of women (predictor) with poor outcomes (dependent variable) (total effect), and (4) the insignificant association of women (predictor) with poor outcomes (dependent variable) when additionally adjusted for failed recanalization (direct effect). Then, using 5,000 bootstrap samples, the significance of the indirect effect was assessed as recommended for small sample sizes [24]. Adjusted odds ratios (aORs) and 95% confidence intervals (CIs) were obtained. All statistical tests were two-tailed, and P-values <0.05 were considered statistically significant.
RESULTS
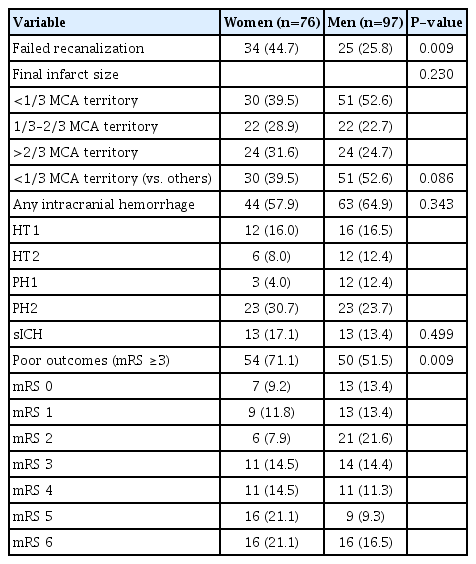
Among 173 patients, 76 were women and 97 were men. Compared to men, women were older and tended to have less premorbid independence and more frequent antiplatelet medication use. Women more frequently presented with atrial fibrillation and were more likely to have a cardioembolic or undetermined stroke than men. Men were more likely to be current smokers and tended to have higher pretreatment CTA-SI ASPECTS scores than women (Table 1). Women differed from men in terms of some clinical outcomes. In women, failed recanalization was more common, and there was a statistical trend toward a larger final infarct size. Above all, women were more likely to have poor outcomes (mRS ≥3) than men, although there was no significant difference in intracranial hemorrhage between sexes (Table 2).
Women were associated with poor functional outcomes in the univariate analysis (Table 3). However, when age and all variables with P<0.1 in the univariate comparison were included in the multivariable logistic regression analysis, sex was no longer associated with functional outcome (result C’ in Fig. 1). Mediation analysis using Baron and Kenny’s causal steps approach was performed to test if failed recanalization mediated the association between sex and functional outcome. All analyses were adjusted for age and variables with P<0.1 in the univariate analysis for each dependent variable. Therefore, in the analysis wherein poor outcomes were the dependent variable, age, premorbid independence, previous anticoagulant use, NIHSS score at admission, M2 occlusion (vs. internal carotid artery or M1 occlusion), CTA-SI ASPECTS, and poor collateral status were adjusted (Table 3). In addition, in the analysis wherein failed recanalization was the dependent variable, age, systolic blood pressure at admission, NIHSS score at admission, CTA-SI ASPECTS, and poor collateral status were adjusted (Supplementary Table 1). These four steps are listed in Fig. 1. (1) The effect of female sex on failed recanalization was significant (A). (2) The effect of failed recanalization on poor functional outcomes adjusted for female sex was significant (B). (3) The total effect of female sex on poor outcomes was significant (C). (4) The direct effect of female sex on poor outcomes adjusted for failed recanalization was non-significant (C’). Moreover, the indirect effect between female sex and poor outcomes via failed recanalization was statistically significant (A×B).
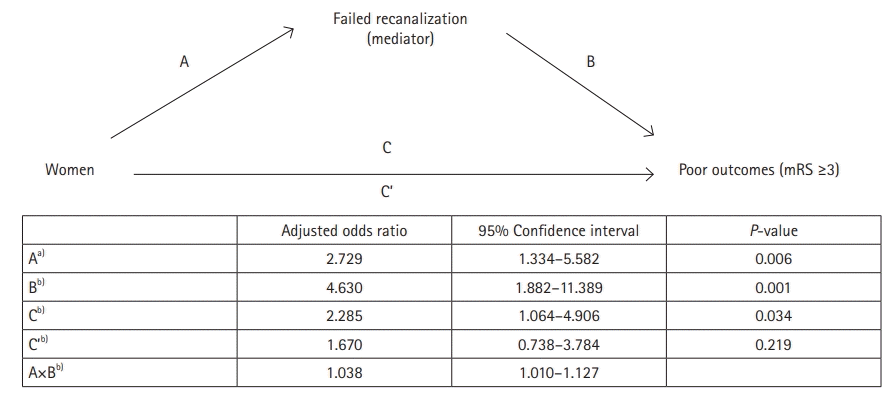
Mediation analysis: failed recanalization as a mediator for the association of women with poor outcomes. mRS, modified Rankin Scale; A, the effect of women on failed recanalization; B, the effect of failed recanalization on poor outcomes additionally adjusted for women; C, total effect of women on poor outcomes; C’, direct effect of women on poor outcomes additionally adjusted for failed recanalization; A×B, indirect effect between women and poor functional outcomes via failed recanalization. All analyses were adjusted for age and variables with P<0.1 in the univariate analysis for each dependent variable. a)Adjusted for age, systolic blood pressure at admission, National Institutes of Health Stroke Scale score at admission, CT (computed tomography) angiography source image Alberta Stroke Program Early CT Score (CTA-SI ASPECTS), and poor collaterals; b)Adjusted for age, premorbid independence, previous anticoagulant use, National Institutes of Health Stroke Scale score at admission, M2 occlusion (vs. internal carotid artery or M1 occlusion), CTA-SI ASPECTS, and poor collaterals.
DISCUSSION
Failed recanalization as a mediator of poor outcomes in women
Similar to previous studies [7,11], women were older and presented with atrial fibrillation more frequently than men. Notably, women were more likely than men to have poor functional outcomes at 3 months. The association remained significant even after adjusting for several confounders, but became statistically insignificant when the recanalization outcome was additionally adjusted. However, it is incorrect to conclude that there was no association between sex and functional outcomes in our study. Instead, as proven by the mediation analysis, it is reasonable to conclude that women were associated with failed recanalization, which mediated the association between sex and poor outcomes.
Sex differences in thrombectomy outcomes: a short literature review
There are many reports in the literature regarding sex differences in thrombectomy outcomes. However, the results of these studies have been conflicting. Several results were obtained from clinical trials that tested thrombectomy. An analysis of 500 patients from an RCT of thrombectomy in the Netherlands (Multicenter Randomized Clinical trial of Endovascular treatment for Acute ischemic stroke in the Netherlands) showed a significant interaction between sex and thrombectomy treatment (P=0.016) with higher mortality in women than in men [8]. Secondary analysis of 182 patients from the multicenter randomized controlled trial of endovascular therapy following imaging evaluation for ischemic stroke (DEFUSE 3) trial data also demonstrated a statistical trend for a lower 90-day functional independence in women than in men within the thrombectomy arm (35% vs. 54%, P=0.059) [9]. However, no difference in outcome between sexes was reported in the analyses based on a larger number of clinical trial patients. The pooled analyses of 1,762 patients from seven RCTs within the Highly Effective Reperfusion evaluated in Multiple Endovascular Stroke Trials (HERMES) collaboration reported no difference in functional independence (mRS 0-2) at 90 days between men and women (48% vs. 48%) treated with thrombectomy [6]. An analysis of 389 patients from the three clinical trials (SWIFT, STAR, and SWIFT PRIME) also showed a similar rate of functional independence at 90 days between sexes (aOR, 1.01; 95% CI, 0.64–1.59) despite older ages and a higher rate of atrial fibrillation for women [7]. Uniquely, a recent study of 198 patients from a multi-center prospective cohort study data (CRISP) reported a favorable shift in the mRS score (aOR, 1.79; 95% CI, 1.04–3.08) with better collaterals and less ischemic core growth (median, 15 mL vs. 29 mL; P<0.01) in women [25].
Meanwhile, most studies based on nonclinical trial data have suggested worse thrombectomy outcomes in women than in men, which is similar to our study. A study of 2,420 patients with large vessel occlusion from a Japanese multi-center registry showed a tendency for poor outcomes among women in both groups who underwent thrombectomy (aOR, 0.83; 95% CI, 0.63–1.09) and who did not undergo thrombectomy (aOR, 0.73; 95% CI, 0.52–1.04) [26]. A single-center study of 279 patients reported lower 90-day functional independence in women (aOR, 0.37; 95% CI, 0.16–0.87) even after adjustment for confounders [12]. Similarly, a study of 2,316 patients from the German registry revealed higher mortality (30.7% vs. 26.4%) with less functional independence (33.2% vs. 40.6%) at 90 days in women. However, this association disappeared after adjusting for confounders [11]. Apart from the above studies, a single-center study of 145 patients reported no significant difference in 3-month functional independence between men and women (60.9% vs. 66.7%) [10].
Possible reasons for more failure of recanalization in women
To sum up the aforementioned studies, there appears to be a trend toward poor functional outcomes in women after thrombectomy under certain circumstances. However, most of the studies described no sex difference in recanalization outcome during thrombectomy [6-11,25]. In this regard, their results are different from ours, indicating poorer outcomes in women mediated by failed recanalization. In fact, only a few reports to date have shown a more frequent failure of recanalization in women during thrombectomy [27,28]. The exact reason for the higher rate of failed recanalization among women in our study is presently unknown. Instead, there is only a speculative presumption. That is to say, sex disparities in arterial diameter might be responsible for the sex difference in recanalization outcome [27,29]. Reportedly, the mean diameters of the terminal internal carotid artery and M1 were smaller in women than in men [29,30]. As the flow rate is proportional to the vessel radius to the fourth power according to the Hagen-Poiseuille equation, the diameter of the treated large arteries or collateral vessels could be a crucial factor determining the blood flow directed to the ischemic area. In women, a smaller arterial diameter can be a disadvantageous factor for reperfusion and functional outcomes in patients undergoing thrombectomy [29]. Similarly, the relationship between vessel diameter and procedural outcomes was reported in a study of 254 patients undergoing endovascular treatment for iliac artery diseases. The investigators showed that the target vessel diameter measured using CT angiography was an independent predictor of procedural failure or vessel-specific complications [31].
On the other hand, sex-specific differences in intravascular coagulation and fibrinolysis could be responsible for the more frequent failure of recanalization in women. A study of South Asian stroke patients reported higher plasminogen activator inhibitor-I and factor VII levels in women [32], which suggests a decreased fibrinolytic potential and increased coagulation tendency leading to a reduced potential of reperfusion in women during thrombectomy.
Limitations and strengths
Our study had several limitations. Above all, this was a single-center retrospective study with a small sample size, thus potentially producing a selection bias. Therefore, our results might be explained by chance and could not be generalized to other stroke populations. In addition, we could not analyze several factors affecting clinical outcomes, such as psycho-cognitive morbidities and social factors. For example, we had no information about the cohabitation of family members, although less family support may be associated with the time taken to arrive at the hospital and post-stroke functional recovery [33]. Generally, women are more likely than men to be widowed and living alone. Therefore, such uncorrected factors could intensify the deviation toward a poorer functional outcome in women [34]. Presumably, the lower proportion of women than men included in our study suggests their later arrival beyond the time window for thrombectomy due to the absence of cohabitants. This is similar to why IVT is less frequently used in women than in men [34-36]. In addition, we had no data on arterial diameter (as discussed), thrombus composition, and vascular access problems that could affect recanalization outcomes [37].
In contrast, our study has strengths in that it adds to real-world data on sex differences in stroke outcomes after thrombectomy. Clinical trial conditions are different from those of real-world clinical practice [38]. Clinical trials are designed to include patients who are most likely to benefit from the tested treatment and to exclude those who are likely to be complicated by the treatment. However, in real-world clinical practice, many emergent large-vessel occlusion patients with adverse conditions, such as low ASPECTS scores, pre-stroke disability, and multiple comorbidities, are treated with thrombectomy because there is no better option [38].
Conclusions
In our stroke patients treated with thrombectomy, female sex was associated with failed recanalization, which mediated the association of female sex with poor functional outcomes. However, this might be explained by chance given our limited study population.
Notes
Ethics statement
This study was approved with a waiver of informed consent by the Institutional Review Board of Soonchunhyang University Bucheon Hospital (No. SCHBC 2021-04-018).
Conflict of interest
No potential conflict of interest relevant to this article.
Author contributions
Conceptualization: SJL. Data curation: SJL, TKL. Formal analysis: SJL, JEM. Investigation: SJL, TKL. Supervision: SJL. Visualization: SJL. Writing–original draft: SJL, JEM. Writing–review & editing: all authors.
Supplementary materials
Supplementary materials can be found via https://doi.org/10.18700/jnc.220054.
Comparison of characteristics between patients with failed and successful recanalization
jnc-220054-suppl.pdf